LabWare LIMS-ELN
A Laboratory Information Management System (LIMS) objective is to assist the laboratory in managing its samples, test results, and associated data and improve its overall productivity.
The LIMS helps laboratories standardize their workflows, tests, and procedures while providing accurate process controls.
The recent versions of LIMS solutions have a configurable and adaptable software design, which expedites the implementation procedures, reduces costs, and minimizes the risk of obsolescence.
KDE Group is LabWare's representative in Israel, specializing in Laboratory informatics management systems for laboratories from all industries.
Our clients come from a wide range of sectors,Pharmaceutical, Healthcare, CBD/THC(Medical Cannabis), Bio-banking, Biology and Clinical,Cosmetics,Farming, AgroChem, Food-Tech,Food and Beverage, Environmental, Forensics, Contract Services,Oil and Gas,Process and Chemical, Avionics, and more
As the World-leading LIMS software company, LabWare provides laboratory automation and solutions suite to more than 29,000 Laboratories from 125 countries.
Our clients produce over a billion samples Per Year, and 98% of customers Recommend Labware.
LabWare's Laboratory Automation system is the most functional, configurable, easily integrated solution, delivering the most value to customers for current and future-focused needs.
LabWare's LIMS follows FDA guidelines and industry best practices (GMP, GLP), and can also work with an electronic lab notebook (ELN), for organizing lab experiments.
LabWare's Top LIMS features include :
Lot Management / Batch Manager - LabWare LIMS organizes batches and lots' tracking, testing, and reporting.
Stability Management - Flexible Stability Management coordinates and manages inventory and storage location.
Environmental Monitoring - Flexible and reliable controls.
Inventory management - Track quantity, location, expiry date, and vendor information for laboratory items such as chemical and consumables
Instrument Interfacing - High capabilities for any instrument interfacing.
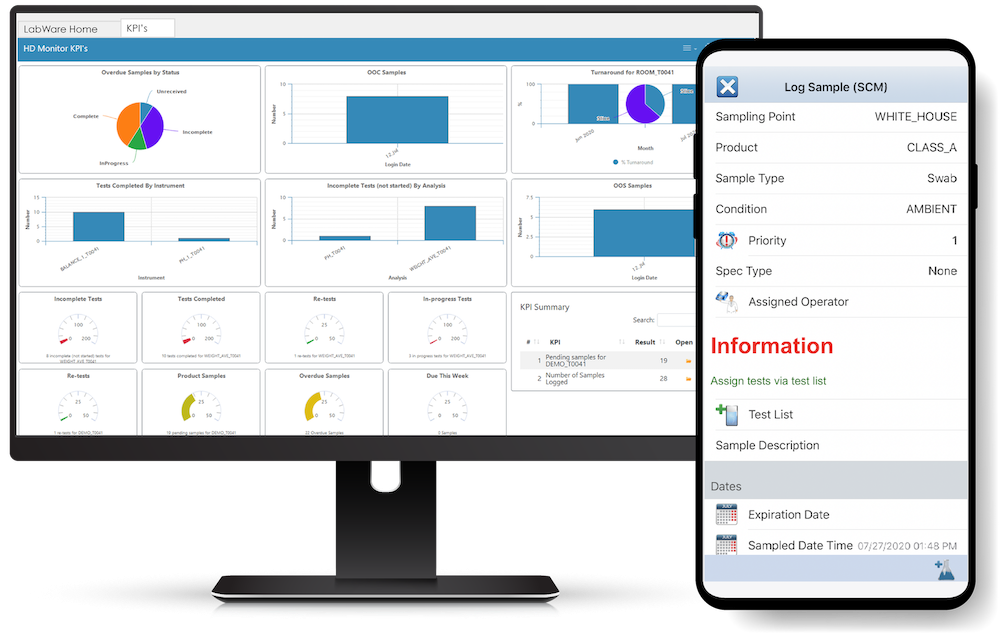
Sample Login / Management - Track your sample management easily and digitally.
Workflows and Dashboards - Advanced search and presentation capabilities for any laboratory personnel.
LabWare LIMS System improves laboratory productivity, throughput, efficiency, data integrity, and compliance, by bringing you the technology for the Future.
LabWare's unique technology ensures the perennity and upgradability of our customer's systems.
The system is constantly enriched based on our customers' requests and needs and as such, focused only on value-added system upgrades.
Request DemoWHAT'S NEW AT LABWARE?
LabWare 8 is the latest unique Enterprise Laboratory Platform, combining the capabilities of a Laboratory Information Management System (LIMS) and an Electronic Laboratory Notebook (ELN) in a single comprehensive solution. As a result, LabWare 8 is the most adaptable and functionally complete LIMS system and offers a proven solution for any size laboratory.
The Platform can suit Customers ranging from a single site, single user implementations to global organizations of all sizes. In addition, LabWare 8 offers new levels of system performance, features for comprehensive compliance with Regulatory Data Integrity mandates, and a wide range of new functions that improve both the end-user experience and system administrative tasks.
LabWare's Mobile App
LabWare MOBILE allows users to connect easily to their LabWare systems, see data and perform customer-defined tasks while trusting the security features applied throughout the LabWare platform.
The LabWare MOBILE app complements LabWare's Laboratory Information Management System (LIMS) and can be used to perform typical LIMS functions such as:
- Sample Login
- Sample Receipt
- Test Assignment
- Result Entry
- Data Reviewing
- Reporting
- Instrument Management
- And more!
LabWare MOBILE is connected to your company's LabWare server through Wi-Fi or cellular data network and is configured to work on both Android and iOS devices.
LabWare Mobile additional features allow
- Creating and uploading files and photos
- Using the mobile camera for barcode scanning and QR codes
- GPS/Location and interface to Mapping App
- PDF viewing
- Date spinners, keypad, keyboard, autocorrect, dictation, etc.
Covid 19
LabWare's contribution to the Covid 19 global fighting:
LabWare is a crucial partner in helping public and private laboratories worldwide in their response to the COVID 19 pandemic.LabWare has been working with public and private sector organizations with its knowledge and advanced technology to implement COVID-19 testing capabilities, advance workflow, and operational efficiency enhancements to increase laboratory testing capacity to meet the unprecedented testing demands.
Labware contributions include assisting public health customers around the world, in implementing new COVID-19 tests and communications protocols for reporting to key regulators players, including the American Center for Disease Control (CDC).
In Israel, KDE Group provided immediate response to the strong requirements of the Israeli Minister of Health, by implementing Labware's LIMS, in the major Covid 19 laboratory. KDE and LabWare were able to support the constant demand and to face the magnitude waves of the pandemic, analyzing over 100,000 tests per day.
LabWare SAAS LIMS
Saas - Software as a service (also known as on-demand software, Web-based/Web-hosted software) is a software service given on a subscription basis. The software is located on cloud infrastructure and is available from any device with an internet connection and or web browser.
Labware Saas LIMS is a powerful and affordable LIMS; it preserves budgets, minimizes IT infrastructure costs, and eliminates future upgrade costs.
The pre-configured SaaS LIMS can be installed in less than 30 days (about 4 and a half weeks). Moreover, with faster Throughput, better Data Integrity/Security (SOC-2 certified and aligned with the latest regulatory data integrity guidelines), the system will advance your overall outcome.
LabWare SAAS LIMS will protect your organization from the threat of failed audits, cyber-attacks, and system downtime.
The system complies with the latest regulatory requirements, such as ISO 17025 and 21 CFR Part 11.
Health & Safety Management
For the crucial Health & Safety of your employees, the EHS management system will track, report, and analyze trends of all safety incidents and meet compliance requirements.
The system offers mobile applications that can be used across the whole organization. KDE will drive the process of fitting the out-of-the-box solutions to fit your company's specific needs and requirements.
Quality Management
KDE's implementation of integrated Quality Management Software (QMS) will improve your company's overall outcome. Decision making is the result of secured and centralized data, which will help cost reduction, and business improvement.
Our solutions for improvement of your Planning and Process QMS integrated system will improve your working plan by adjusting to your workflows and will manage your procedures. Our QMS is following FMEA, APQP, and PPAP regulations.
Our solution for managing non-conformances QMS integrated system will inspect, capture, and report any relevant data, internal/external, and non-conformances while producing corrective and preventative actions in response.
Environmental Management
KDE's implementation of Environment Management Software (EMS) will assure your company's environmental compliance and will enable sustainability reporting for air emissions, water quality metrics, soil contamination, and hazardous waste tracking.
Our solution for Smart Reporting The EMS follows global regulatory reporting forms. Files are easy to access, and your environmental data is constantly updated and ready to be submitted.
Our solution for Measuring & Monitoring The EMS will track, consolidate, and report all environmental data, air, water, waste emissions from any site, location, or source.
ALDON
We provide solutions for Software Lifecycle Management and Service-oriented Architecture (SOA) based on Rocket Software's ALDON system. The system assures regulatory compliance and monitoring tools for business processes, with information security using the computing resources infrastructure of TANGO/04.
PKWARE
KDE also represents PKWARE worldwide known Productivity and Protected software solutions. The award-winning provider of data security solutions incorporating data discovery, classification, and protection in automated, policy-driven workflows for enterprises worldwide. PKWARE invented the ZIP file format and has a 30+ year legacy of innovation. PKWARE off-the-shelve tools are the most comprehensive enterprise data protection suites ever created, allowing businesses to eliminate security gaps, take control of sensitive data and meet the most stringent data compliance requirements. Unmatched security, ease of use, and rule-based centralized control make PKWARE the choice for enterprise data protection. It includes predefined, out-the-box policies that enable organizations to begin classifying data quickly with just a few clicks. It includes Data Discovery, Classification, Encryption & Masking, Monitoring, Reporting and Automation.
Software Diversified Services
KDE is the arm of Software Diversified Services - USA. We enable the enterprise customer to See what's happening on the network anywhere, any time. VIP product monitors the IP network, Enterprise Extender, OSA, FTP and prepares network history reports. It verifies service-level agreements, too. VIP can trigger automatic repairs by sending alerts to its companion product, VitalSigns for Network Automation and Control (VNAC). Detect, diagnose, resolve, and prevent mainframe TCP/IP problems on z/OS networks. The user has real-time graphic displays of z/OS TCP/IP network health and traffic. VIP gives total NETWORK VISIBILITY into the graphic network from a single desktop browser interface, monitoring all corners of the system including remote applications.